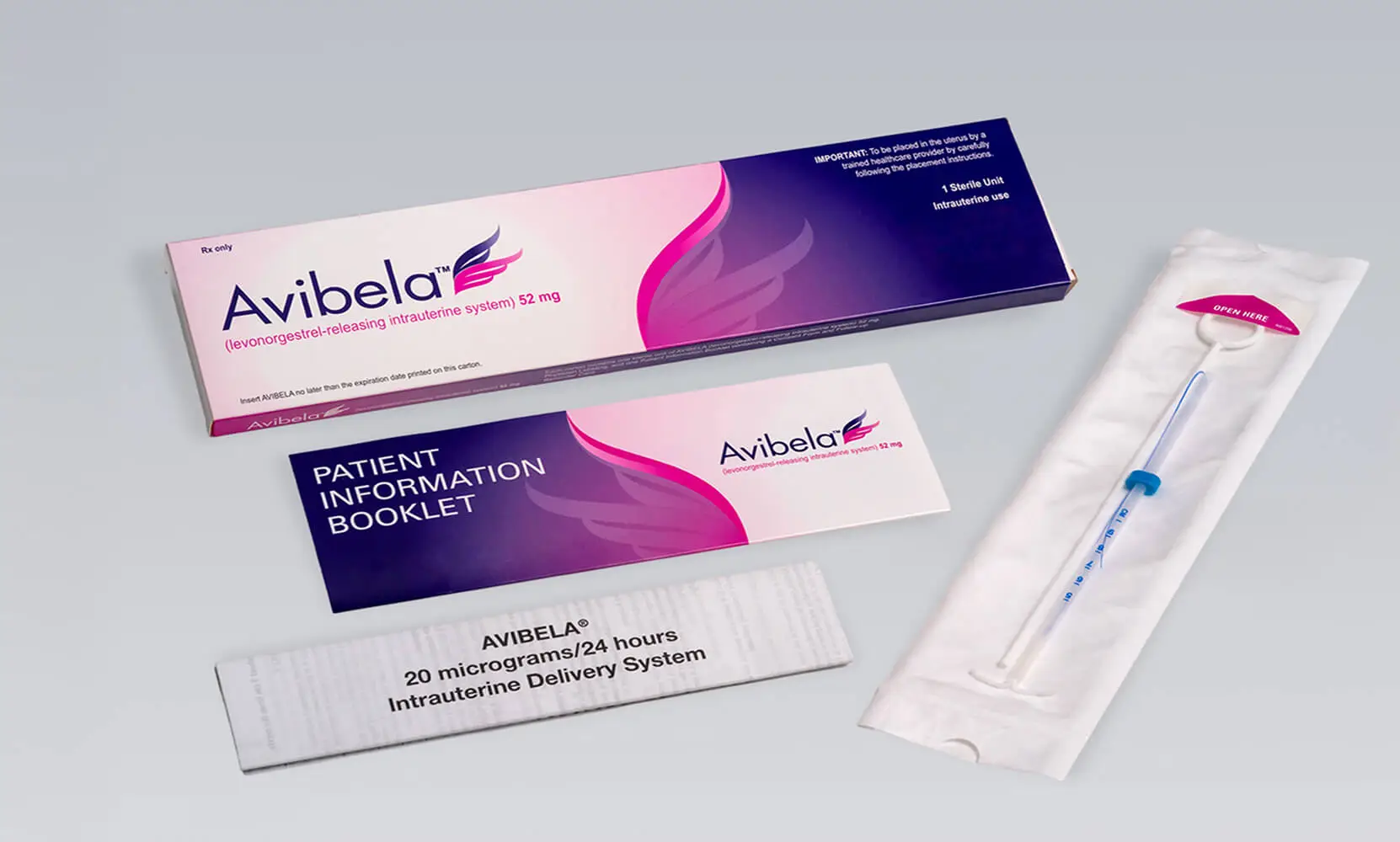
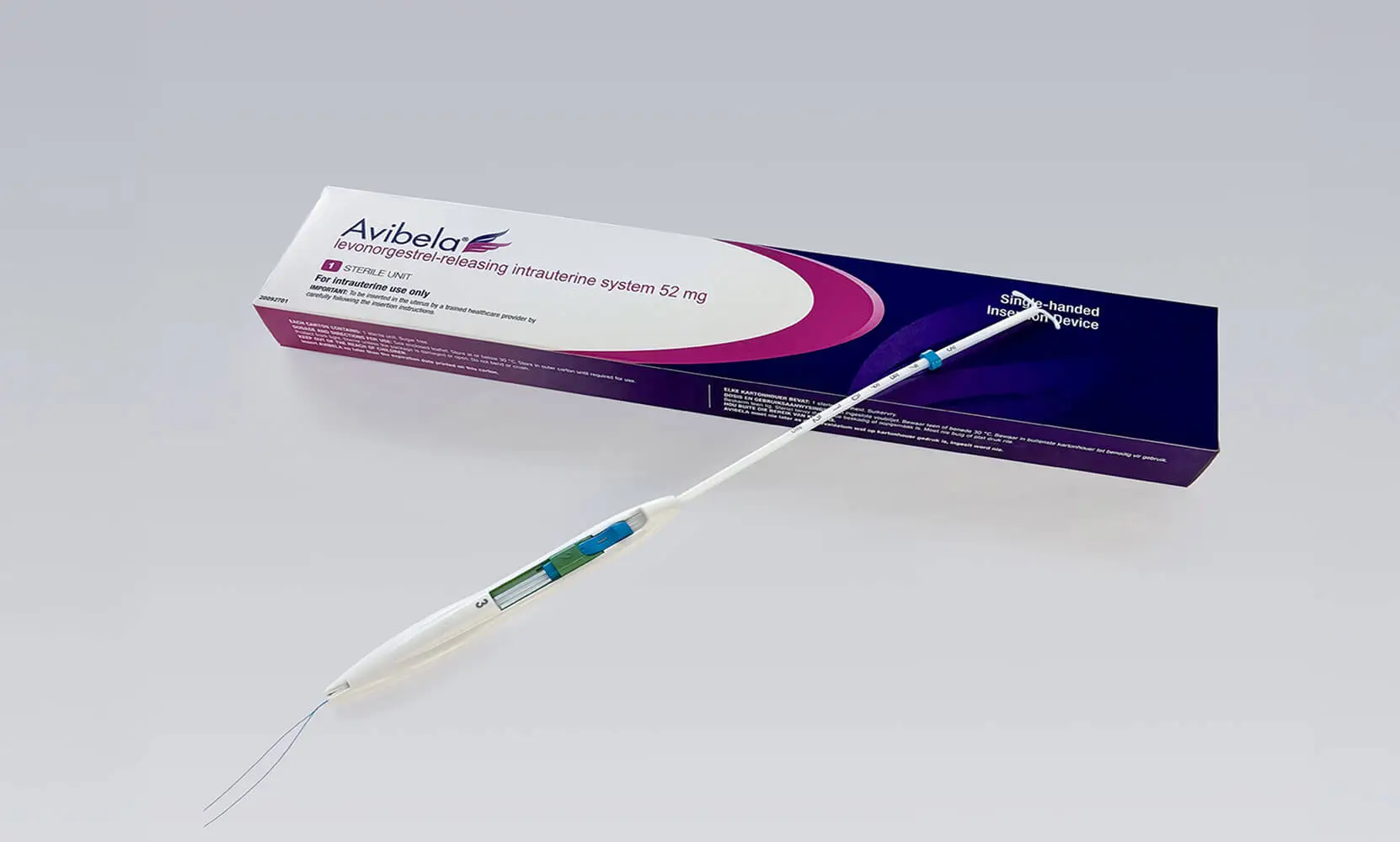
Avibela® levonorgestrel-releasing intrauterine system, 52 mg
Indicated to prevent pregnancy for up to 8 years and to treat heavy menstrual bleeding for up to 5 years
Key Clinical Characteristics
Highly effective, long-acting reversible contraception
Highly effective, long-acting reversible contraception
About 99% effective at preventing pregnancy for up to 8 years
Studied in a broad range of women (ages 16–45; BMI range 16–62; 58% nulliparous)
Treats heavy menstrual bleeding (HMB)
Treats heavy menstrual bleeding (HMB)
Effective treatment in 80% of women with HMB
Reduces menstrual blood loss by 96% after 6 months
Can be inserted on the same day it is prescribed
Can be inserted on the same day it is prescribed
At any time during the menstrual cycle in non-pregnant women
Before STI test results in women with no clinical evidence of infection
Quick return to fertility after removal
Quick return to fertility after removal
Women can become pregnant immediately after IUD removal
Fertility rates within one year of removal are consistent with the general population
See Important Safety Information
Expanding Equitable Access to the Hormonal IUD around the Globe
AVIBELA can be made available in 88 countries across Africa, Central America and the Caribbean, and South and Southeast Asia. Download approved product labeling for each country by region below.
SMPC
Summary of Product Characteristics
PIL
Patient Information Leaflet

Africa
Africa
-
Import waiver required
BurundiCameroonCape VerdeCentral Afr. RepublicComorosChadDjiboutiEquatorial GuineaEgypt -
Import waiver required
EritreaEthiopiaGabonGhanaGuineaGuinea-BissauIvory CoastLesothoLiberia -
Import waiver required
LibyaMaliMauritaniaMauritiusMayotteMoroccoNamibiaNigerRepublic of Congo -
Import waiver required
Sao Tome & PrincipeSenegalSeychellesSierra LeoneSomaliaSouth SudanSudanSwazilandThe Gambia -
Import waiver required
TogoTunisia

South & Southeast Asia
South & Southeast Asia
Import waiver required
-
BangladeshBhutanCambodiaIndiaIndonesiaLao PDRMalaysiaMaldivesMyanmarNepalPakistanPapua New GuineaPhilippinesSri LankaThailand
-
Timor-LesteVietnam

Central America and the Caribbean
Central America and the Caribbean
Import waiver required
-
BelizeCosta RicaCubaDominiciaDominican RepublicEl SalvadorGrenadaGuatemalaHaitiHondurasJamaicaNicaraguaPanamaSt. Kitts and NevisSt. Lucia
-
St. Vincent & the Grenadines

How to Access AVIBELA
AVIBELA can be ordered from our global distribution partner, DKT WomanCare Global.
For information about how to access AVIBELA in South Africa, please contact Pharma Dynamics.
Interested in learning more about AVIBELA or partnering with us?
Submit your inquiry below and our team will follow up promptly.
Training Materials
Please refer to the approved product labeling in your country for the approved duration of use, as the 8-year duration of use for contraception may still be pending approval in some countries.
Who is not appropriate for AVIBELA
AVIBELA is contraindicated when one or more of the following conditions exist: pregnancy; for use as post-coital contraception (emergency contraception); congenital or acquired uterine anomaly, including leiomyomas, that distorts the uterine cavity and would be incompatible with correct intrauterine system (IUS) placement; acute pelvic inflammatory disease (PID); postpartum endometritis or infected abortion in the past 3 months; known or suspected uterine or cervical malignancy; known or suspected breast cancer or other hormone-sensitive cancer, now or in the past; uterine bleeding of unknown etiology; untreated acute cervicitis or vaginitis, including bacterial vaginosis, known chlamydial or gonococcal cervical infection, or other lower genital tract infections until infection is controlled; acute liver disease or liver tumor (benign or malignant); conditions associated with increased susceptibility to pelvic infections; a previously inserted IUS that has not been removed; a history of hypersensitivity reaction to any component of AVIBELA.
Indications
AVIBELA is a sterile, levonorgestrel-releasing intrauterine system indicated for contraception for up to 8 years; replace after 8 years if continued use is desired. AVIBELA is indicated for treatment of heavy menstrual bleeding (HMB) for up to 5 years; replace after 5 years if continued use is desired. AVIBELA may be particularly useful in women with HMB requiring (reversible) contraception.
Who is not appropriate for AVIBELA
AVIBELA is contraindicated when one or more of the following conditions exist: pregnancy; for use as post-coital contraception (emergency contraception); congenital or acquired uterine anomaly, including leiomyomas, that distorts the uterine cavity and would be incompatible with correct intrauterine system (IUS) placement; acute pelvic inflammatory disease (PID); postpartum endometritis or infected abortion in the past 3 months; known or suspected uterine or cervical malignancy; known or suspected breast cancer or other hormone-sensitive cancer, now or in the past; uterine bleeding of unknown etiology; untreated acute cervicitis or vaginitis, including bacterial vaginosis, known chlamydial or gonococcal cervical infection, or other lower genital tract infections until infection is controlled; acute liver disease or liver tumor (benign or malignant); conditions associated with increased susceptibility to pelvic infections; a previously inserted IUS that has not been removed; a history of hypersensitivity reaction to any component of AVIBELA.
Clinical considerations for use of AVIBELA
Use AVIBELA with caution after careful assessment if any of the following conditions exist and consider removal of AVIBELA if any of them arise during use: coagulopathy or use of anticoagulants; migraine, focal migraine with asymmetrical visual loss, or other symptoms indicating transient cerebral ischemia; exceptionally severe or frequent headache; marked increase of blood pressure; or severe arterial disease such as stroke or myocardial infarction. Consider removal of AVIBELA if the any of the following conditions arise during use: uterine or cervical malignancy, or jaundice. Because irregular bleeding/spotting is common during the first months of AVIBELA use, exclude endometrial pathology (polyps or cancer) prior to the insertion of AVIBELA in women with persistent or uncharacteristic bleeding. If the threads are not visible or are significantly shortened, they may have broken or retracted into the cervical canal or uterus. If AVIBELA is displaced (e.g., expulsed or perforated the uterus), remove it.
Pregnancy related risks with AVIBELA
If pregnancy should occur with AVIBELA in place, attempt to remove AVIBELA because leaving it in place may increase the risk of spontaneous abortion and preterm labor. Removal of AVIBELA or probing of the uterus may also result in spontaneous abortion. Evaluate for ectopic pregnancy because the likelihood of a pregnancy being ectopic pregnancy is increased. Tell people about the signs of ectopic pregnancy and associated risks, including loss of fertility. People with a history of ectopic pregnancy, tubal surgery, or pelvic infection have a higher risk of ectopic pregnancy.
Educate about PID or endometritis
Insertion of AVIBELA is contraindicated in the presence of known or suspected PID or endometritis. IUSs have been associated with an increased risk of PID, most likely due to organisms being introduced into the uterus during insertion. In the contraception study, one person diagnosed with PID and two people diagnosed with endometritis developed the infection within a week of insertion. One endometritis case was diagnosed at 39 days after insertion. The remaining 11 cases of PID and endometritis were diagnosed more than 6 months after insertion, including one at 30 days after IUS removal. In the HMB study, one person was diagnosed with PID about 5 months after insertion. Counsel people who use AVIBELA to notify a healthcare provider if they develop lower abdominal or pelvic pain, fever, chills, unusual or malodorous discharge, unexplained bleeding, genital lesions or sores, or dyspareunia. PID and endometritis are often associated with sexually transmitted infections (STIs); AVIBELA does not protect against STIs, including HIV. PID or endometritis may be asymptomatic but still result in tubal damage and its sequelae. Inform people about the possibility of PID or endometritis and that these infections can cause tubal damage leading to ectopic pregnancy or infertility, or infrequently can necessitate hysterectomy, or cause death.
Expect changes in bleeding patterns with AVIBELA
Spotting and irregular or heavy bleeding may occur during the first 3 to 6 months. Periods may become shorter and/or lighter thereafter. Cycles may remain irregular, become infrequent, or even cease. Consider pregnancy, including ectopic pregnancy, if menstruation does not occur within 6 weeks of the onset of previous menstruation. If a significant change in bleeding develops during prolonged use, conduct diagnostic tests to assess possible endometrial pathology.
Be aware of other serious complications and most common adverse reactions
Some serious complications with IUSs like AVIBELA are sepsis, perforation, and expulsion. Severe infection or sepsis, including Group A streptococcal sepsis (GAS), have been reported following insertion of other LNG-releasing IUSs. Aseptic technique during insertion of AVIBELA is essential to minimize serious infections such as GAS.
Perforation (total or partial, including penetration/embedment of AVIBELA in the uterine wall or cervix) may occur, most often during insertion, although the perforation may not be detected until sometime later. Perforation may also occur at any time during use. Perforation may reduce contraceptive efficacy. If perforation is suspected, locate and remove AVIBELA as soon as possible. Surgery may be required. Delayed detection or removal of AVIBELA in case of perforation may result in migration outside the uterine cavity, adhesions, peritonitis, intestinal perforations, intestinal obstruction, abscesses, and erosion of adjacent viscera. The risk of perforation is increased if inserted in patients who have fixed retroverted uteri, are postpartum, or are lactating. Delay AVIBELA insertion a minimum of 4 weeks or until uterine involution is complete following a delivery or a second-trimester abortion.
Partial or complete expulsion of AVIBELA may occur, resulting in the loss of contraceptive protection. Expulsion risk is increased when inserted immediately after delivery; it appears to be increased with insertions after second-trimester abortion, based on limited data. Risk of expulsion is increased for people with a history of HMB or greater than normal BMI at the time of insertion. Remove a partially expelled AVIBELA. If expulsion has occurred, a new AVIBELA may be inserted when there is reasonable certainty the person is not pregnant.
Ovarian cysts may occur and are generally asymptomatic. Cysts may be accompanied by pelvic or abdominal pain or dyspareunia. Evaluate persistent ovarian cysts.
In the AVIBELA contraception study, the most common adverse reactions (≥10% users) were vulvovaginal mycotic infections, vaginal bacterial infections, acne, nausea or vomiting, abdominal discomfort or pain, and procedural bleeding). In the AVIBELA HMB study, the adverse reaction profile was consistent with the adverse reaction profile in the contraception study.
Teach people to recognize and immediately report signs or symptoms of the aforementioned conditions. Consider evaluating people 4 to 6 weeks after AVIBELA insertion and during routine care, or more often if clinically indicated. Check threads during each evaluation.
Do not use AVIBELA if you:
- are or might be pregnant; AVIBELA cannot be used as an emergency contraceptive
- have a serious pelvic infection called pelvic inflammatory disease (PID); PID occurs in less than 1% of users
- have an untreated lower genital infection now
- have had a serious pelvic infection after an abortion or pregnancy within the last 3 months
Do not use AVIBELA if you:
- are or might be pregnant; AVIBELA cannot be used as an emergency contraceptive
- have a serious pelvic infection called pelvic inflammatory disease (PID); PID occurs in less than 1% of users
- have an untreated lower genital infection now
- have had a serious pelvic infection after an abortion or pregnancy within the last 3 months
- can get infections easily. For example, if you:
- have problems with your immune system
- have multiple sexual partners or your partner has multiple sexual partners
- have a history of PID
- have or suspect you might have cancer of the uterus or cervix
- have bleeding from the vagina that has not been explained
- have short-term (acute) liver disease or liver tumor
- have breast cancer or any other cancer that is sensitive to progestin (a female hormone), now or in the past
- have an intrauterine system in your uterus already
- have a condition of the uterus that changes the shape of the uterine cavity, such as large fibroid tumors
- are allergic to levonorgestrel, silicone, polyethylene, or barium sulfate
Call your healthcare provider right away if you think you are pregnant. If you get pregnant while using AVIBELA, you may have an ectopic pregnancy, which means that the pregnancy is not in the uterus. Unusual vaginal bleeding or abdominal pain, especially with missed periods, may be a sign of ectopic pregnancy. Ectopic pregnancy can cause internal bleeding. There are also risks if you get pregnant while using AVIBELA and the pregnancy is in the uterus. Severe infection and premature labor or delivery can occur with pregnancies that continue with an IUS.
Although uncommon, pregnancy while using AVIBELA can be life-threatening and may result in loss of pregnancy or fertility.
Life-threatening infection can occur within the first few days after AVIBELA is placed. Call your healthcare provider immediately if you develop severe pain or fever shortly after AVIBELA is placed.
Some IUS users get a serious pelvic infection called pelvic inflammatory disease (PID) or endometritis. These may be sexually transmitted. You have a higher chance of getting PID or endometritis if you or your partner has sex with other partners. PID or endometritis can cause serious problems, such as infertility, ectopic pregnancy, or pelvic pain that does not go away. More serious cases of PID or endometritis may require surgery. Removal of the uterus (hysterectomy) is sometimes needed. In rare cases, infections that start as PID or endometritis can even cause death. Tell your healthcare provider right away if you have any of these signs of PID or endometritis: long-lasting or heavy bleeding, unusual or foul-smelling vaginal discharge, low abdominal or pelvic pain, painful sex, genital lesions or sores, chills, or fever.
AVIBELA may partially go into (become embedded) or go completely through (perforate) the wall of the uterus. If perforation occurs, AVIBELA may move outside the uterus and can cause internal scarring, infection, or damage to other organs. You may need surgery to have AVIBELA removed if it is embedded or perforation occurs. Risk of perforation is increased in breastfeeding people.
AVIBELA may come out of your uterus (expulsion). Expulsion occurs in about 4 out of 100 people, most often in the first year of use. You may become pregnant if AVIBELA comes out. If you think that AVIBELA has come out, use another birth control method (like condoms) or do not have sex (vaginal intercourse) until you are seen by a healthcare provider
Ovarian cysts may occur but usually disappear.
Bleeding and spotting may increase in the first 3 to 6 months and remain irregular. Periods over time usually become shorter, lighter, or may stop.
AVIBELA does not protect against HIV or STDs.
If any of the side effects get serious, or if you notice any side effects not listed, please tell your healthcare provider.